 Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Three outcomes have resulted: entropy has increased, some energy has become unavailable to do work, and the system has become less. The result will be water at an intermediate temperature of 30.0 ☌ 30.0 ☌. Robinson, PhD, Richard Langley, Klaus Theopold Suppose we mix equal masses of water that are originally at two different temperatures, say 20.0 ☌ 20.0 ☌ and 40.0 ☌ 40.0 ☌. We recommend using aĪuthors: Paul Flowers, William R. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: except for a perfect crystal at absolute zero, every substance has a finite positive entropy If there is only one possible arrangement of atoms (i.e. The third law of thermodynamics defines absolute zero on the entropy scale. 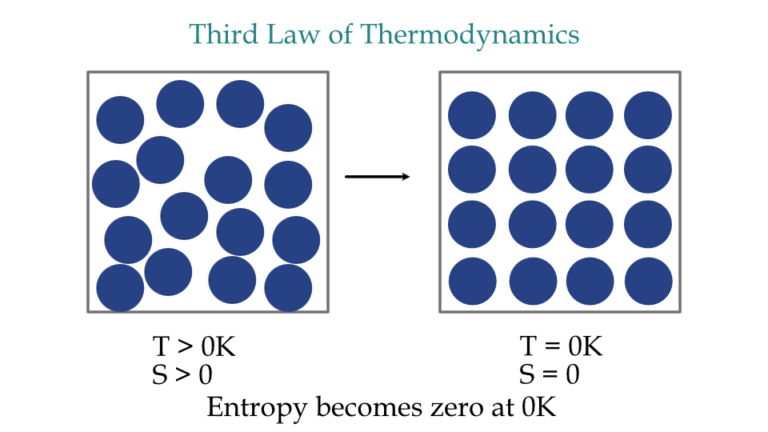 
If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the Fundamental Equations relate functions of state to each other using 1st and 2nd Laws. 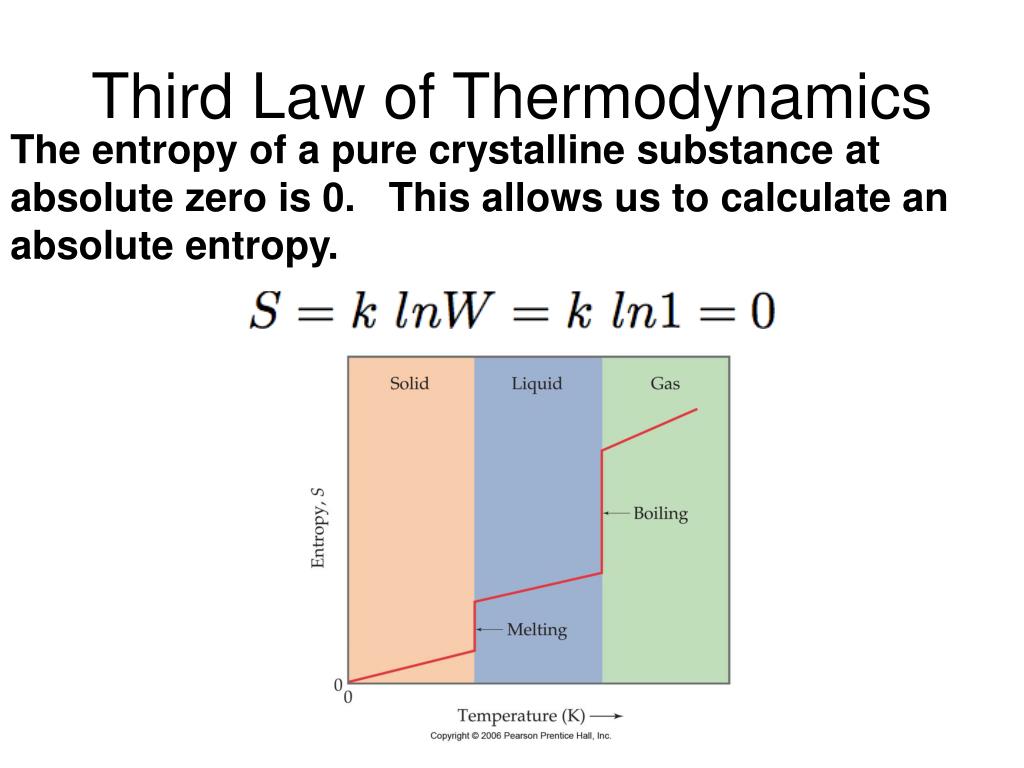
This book may not be used in the training of large language models or otherwise be ingested into large language models or generative AI offerings without OpenStax's permission. Fundamental Equations, Absolute Entropy, and The Third Law. A summary of these three relations is provided in Table 16.1. These results lead to a profound statement regarding the relation between entropy and spontaneity known as the second law of thermodynamics: all spontaneous changes cause an increase in the entropy of the universe. This process involves no change in the entropy of the universe. In this case, the system and surroundings experience entropy changes that are equal in magnitude and therefore sum to yield a value of zero for Δ S univ. The temperature difference between the objects is infinitesimally small, T sys ≈ T surr, and so the heat flow is thermodynamically reversible.This process involves a decrease in the entropy of the universe. We employ classical thermodynamics to gain information about absolute entropy, without recourse to statistical methods, quantum mechanics or the third law of. The third law states that the entropy of a. The magnitude of the entropy change for the surroundings will again be greater than that for the system, but in this case, the signs of the heat changes will yield a negative value for Δ S univ. The Third law of thermodynamics provide information on the calculation of absolute entropy of a system. Finally, substances with strong hydrogen bonds have lower values of S°, which reflects a more ordered structure.The arithmetic signs of q rev denote the gain of heat by the system and the loss of heat by the surroundings. For example, compare the S° values for CH 3OH(l) and CH 3CH 2OH(l). Third Law of Thermodynamics: The entropy of all pure perfect crystalline substances is zero at T 0. Similarly, the absolute entropy of a substance tends to increase with increasing molecular complexity because the number of available microstates increases with molecular complexity. Soft crystalline substances and those with larger atoms tend to have higher entropies because of increased molecular motion and disorder. In contrast, graphite, the softer, less rigid allotrope of carbon, has a higher S° due to more disorder in the crystal. Among crystalline materials, those with the lowest entropies tend to be rigid crystals composed of small atoms linked by strong, highly directional bonds, such as diamond. 
\( \newcommand\) also reveals that substances with similar molecular structures tend to have similar S° values. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
